Controversial Vaccine Trials in Africa Raise Concerns Over US Policy

Experts fear a 'prototype' unethical vaccine trial in Guinea-Bissau may foreshadow studies under RFK Jr's HHS leadership, raising questions about US vaccine policy.
A group of Danish researchers, whose past work on the health effects of vaccines has faced scrutiny, are at the center of a new controversy surrounding a proposed vaccine trial in Guinea-Bissau. Experts fear this trial may serve as a prototype for future studies under the leadership of Robert F. Kennedy Jr., the newly appointed secretary of the US Department of Health and Human Services (HHS) and a longtime critic of mainstream vaccine policies.
The planned trial in Guinea-Bissau would have only vaccinated half of the newborns with the hepatitis B vaccine, despite an 18% prevalence rate of the illness among adults in the region. Hepatitis B can lead to serious and sometimes fatal health consequences. This unethical study design has sparked outrage among global health experts, who warn it could set a dangerous precedent.
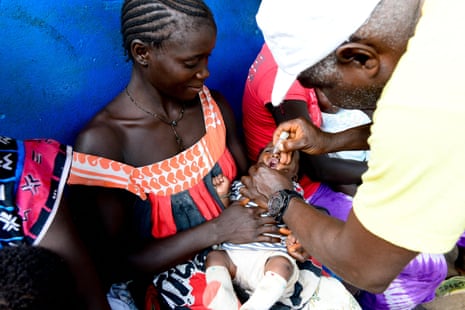
"This is really concerning," said Dr. Sarah Mayanja, a global health researcher. "If this is allowed to move forward, it could pave the way for similar trials in other vulnerable populations, both in Africa and potentially even in the US under RFK Jr.'s leadership at HHS."
The researchers behind the Guinea-Bissau trial have a history of producing studies that challenge the mainstream scientific consensus on vaccine safety and efficacy. Their work has been criticized by the broader scientific community for methodological flaws and potential conflicts of interest.
"These researchers have a track record of producing studies that are misleading and not representative of the broader evidence," said Dr. Emily Chen, an epidemiologist. "Allowing them to conduct trials in vulnerable populations is extremely unethical and could have devastating public health consequences."
Experts fear that if the Guinea-Bissau trial is allowed to proceed, it could set a dangerous precedent that RFK Jr. and the HHS may seek to emulate in the US. This raises serious concerns about the future direction of US vaccine policy under the new administration.
"We can't let this kind of unethical and potentially harmful research become the norm," said Dr. Mayanja. "The stakes are too high, both for the individual participants and for public health more broadly."
As the controversy continues to unfold, global health advocates are calling for strict oversight and rigorous ethical reviews of any proposed vaccine trials, particularly those involving vulnerable populations. The outcome of the Guinea-Bissau case could have far-reaching implications for the future of vaccine research and policy in the US and beyond.
Source: The Guardian


